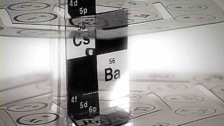
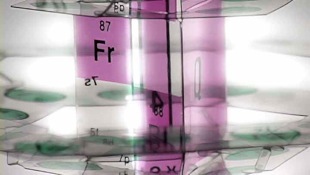
Fernando has created scores of variations on his 3D Periodic Table. Some of them are larger than Fernando.

The 2D Periodic Table can be found in every chemistry lab and classroom in the world. It is the first thing we are taught when we study chemistry.
It is two dimensional.
Fernando Dufour is conceivably the world’s leading authority on the 3D Periodic Table.
The 2D Periodic Table gives you approximately 6 pieces of information per element. Fernando Dufour’s 3D Periodic Tables give you approximately 42 pieces of information per element.
3D makes a big difference.

Photographs of some of Fernando’s models have appeared in prestigious scientific journals and magazines such as Scientific American. In the past his models have always been seen in the context of an ‘intriguing but theoretical or futurist idea’....




“When I first discovered the Periodic Table, I was awed to the ultimate heaven in thinking that this was knowledge so infinite it would unravel all the mysteries of nature – the blueprint of the universe itself.”
“The 2D Periodic Table is now 135 years old. Science has made enormous advances. I found that trying to understand modern concepts in chemistry using a 2D Periodic Table was like trying to fly over the Rockies in a car. You need a plane. You need 3D.”
Fernando Dufour

Recently this has all changed. Here is an excerpt from an article published in numerous prestigious scientific publications. It was written by Eric Scerri, scholar and philosopher and one of the most respected current historians of the Periodic Table:
“Canadian chemist, Fernando Dufour, has produced perhaps the finest 3-D system yet to be devised, called the ElementTree. Its main advantage is the way it highlights secondary periodicity, as in the case of the pyramidal two-dimensional (2-D) table. In the ElementTree, a slide or plane through the 3-D system serves to align elements with similar properties across multiple dimensions. For example, one slide groups many elements showing a maximum valence of +4, such as carbon (C), silicon (Si), germanium (Ge), tin (Sn), titanium (Ti) and zirconium (Zr). The next slide along aligns elements that display a +5 maximum valence, such as phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), vanadium (V) and niobium (Nb). These are patterns hidden within the traditional 2-D periodic table. Until now, the limitations of the 2-D printed page have tended to favour the block form of the periodic table. But clever innovations are giving birth to a plethora of alternative forms, some adding a third dimension. These new forms may be on the verge of making an important contribution to how one of the most important icons in the whole of science – and certainly in chemistry – is displayed. And who knows? In years to come, that ubiquitous wall poster may be replaced by something wildly different, from stacked 3-D tiles to a swirling galaxy.”
Eric Scerri
COSMOS, April/May 2008